Research
We use Drosophila along with a multidisciplinary approach that include in vivo electrophysiology and functional imaging, optogenetics, thermogenetics and behavior experiments to examine how neural circuits encode information from the molecular and cellular level, through the neural circuit level and up to behavioral output. In particular research at lab focuses on the three following questions:
1. The physiological roles of GPCR voltage dependence: over 90% of non-sensory G protein coupled receptors (GPCRs) are expressed in the brain where they play an important role in almost every aspect of brain function.
In the past 20 years, it was demonstrated that the activity as well as agonist binding affinity of several GPCRs is regulated by membrane potential. For example, the activity of cholinergic M2 muscarinic receptors, glutamate receptors mGluR3, and dopaminergic D2 receptor, is reduced by depolarization. In contrast, the activity of M1R, and mGluR1a is increased at positive voltages. However, the physiological roles of this voltage dependency, its effect on neural activity, or its relevance to behavior were never demonstrated.
Recently, we showed that M1 voltage dependence is a crucial component in its recruitments. Not only that under physiological conditions, in vivo, M1 could not be activated without depolarization, but also depolarization alone was sufficient to activate M1. Furthermore, flies with a voltage independent M1 receptor had increased odor habituation indicating a paramount effect on behavior. In addition, preliminary data indicate that voltage dependence of M2 is required for both accurate and specific learning and memory.
We now examine the effects the voltage dependence of dopaminergic, GABAergic and serotonergic GPCRs have on Drosophila learning and memory.

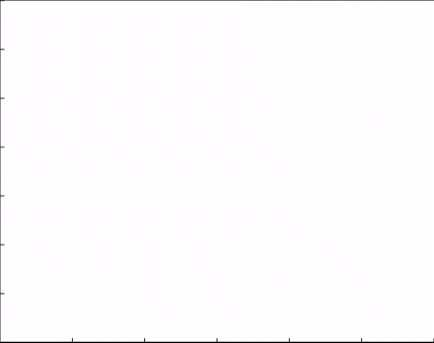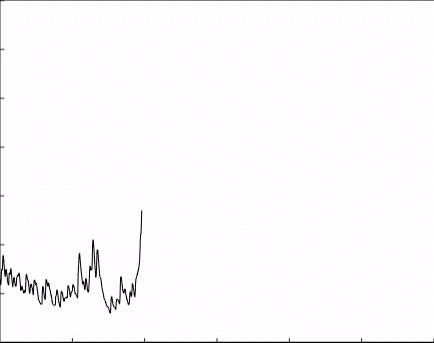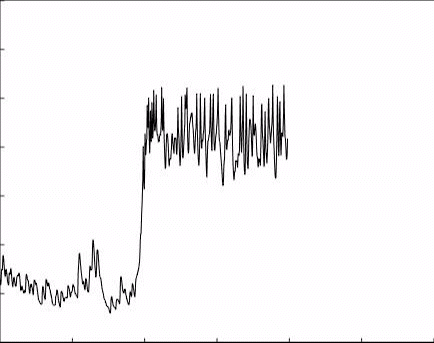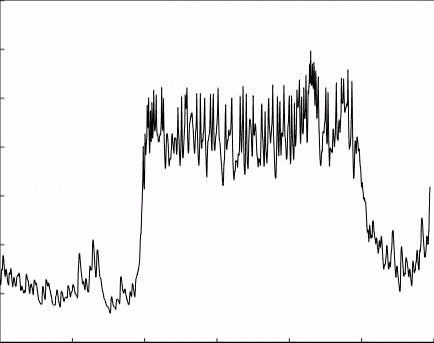

3. GPCR voltage dependence as a therapeutic approach: GPCRs mediate a large variety of physiological events throughout the body and brain, and their role is critical for normal brain functions. Hence, they play a paramount role in therapeutic intervention and are targeted by ∼40% of all presently marketed drugs. Nevertheless, in some cases, no current drug exist that target GPCR involved in the process. For example, in the case of Parkinson’s disease, all available drugs target the levels of the neurotransmitter dopamine. Current methods of drug screening do not address the voltage dependency of the receptors and thus miss potential drugs.
We are developing a novel method for drug screening which directly examines the effects of drugs on the voltage dependence of GPCRs and use this to search for novel therapeutic approaches.



2. Mechanism underlying specific and efficient learning and memory: Associative learning is one of the major mechanisms by which animals learn about their environment. For example a child touching a hot kettle will associate the hot kettle with the induced pain and will refrain from repeating this. Despite years of research on the topic, the mechanisms underlying both specific learning (for example associating a hot kettle but not all kettles with pain), and efficient learning (i.e. learn to completely avoid a hot kettle) are not fully understood. Current models rely on the existence of different types or dopaminergic reward neurons projecting to different brain regions as well as on activity dependent neuromodulation. However the notion of different types or dopaminergic reward neurons projecting to different brain regions requires prior knowledge and scale down the problem of specificity but do not resolve it. The notion of activity dependent neuromodulation is definitely contributing to specificity of learning, but it was shown in many examples that neuromodulation of learning circuits occur even without activity in the target neurons. Thus, while this mechanism is appealing and definitely involved in the formation of specific learning it is not sufficient.
The Drosophila olfactory learning and memory system has all the above components. It has many types of dopaminergic neutrons responding to different signals and projecting to different targets and strong activity dependent neuromodulation. Using this model system we probe the molecular, cellular and circuit components that are required to generate both efficient and specific associative learning.

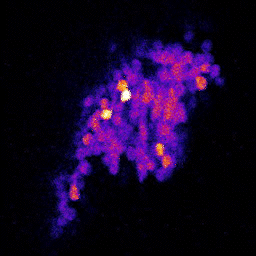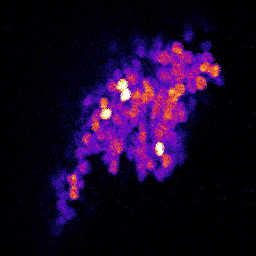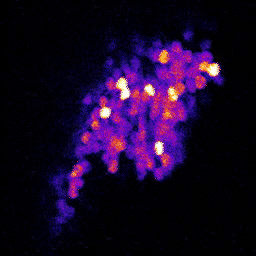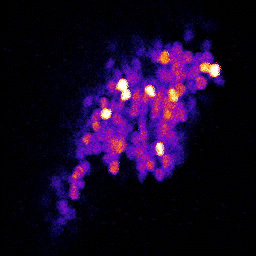
4. Neuronal code reliability in a plastic environment: To survive, animals must recognize reoccurring stimuli. A key requirement for repeated identification of stimuli is reliable representation by the neural code on each encounter. Synaptic transmission underlies neural codes propagation between brain regions. A hallmark of chemical synapses is their plasticity, which enables signal transfer to be modified in an activity-dependent manner. Despite many decades of intense research on synapses, it remains unclear how the plastic features of synaptic transmission can maintain reliable neural coding. By studying the olfactory system of Drosophila melanogaster, we aim to obtain a deeper mechanistic understanding of how synaptic function shapes neural coding reliability in the live, behaving animal.


